Quick Answer
Photoinitiator 1173 (CAS 7473-98-5) is a liquid. Photoinitiator 184 (CAS 947-19-3) is a solid. Both are Norrish Type I alpha-hydroxy ketones with nearly identical UV absorption peaks (320–335 nm) and excellent non-yellowing profiles.
Decision Factor Choose 1173 Choose 184 Production format Liquid mixing systems Solid-fed or high-temp batch Handling complexity Low — no pre-dissolution Higher — dissolve first Migration sensitivity Higher (lower MW) Lower (better for outdoor/weathering) Co-PI solubilization Better as liquid carrier Synergistic when blended 50:50 with 1173 LED UV systems Neither alone — pair with TPO-L Neither alone — pair with TPO-L Neither performs well alone. Both need a co-initiator or blending strategy to address oxygen inhibition and achieve surface cure.
The Problem Nobody Tells You Before You Order
You’re formulating a UV coating. The data sheet says “typical loading: 2–4%.” You pick one, run the line — and the surface stays tacky. Or you get full cure but yellowing appears after two weeks under sunlight. Or your supplier ships 200 kg of solid 184 when your mixing system was built for liquids.
Choosing wrong between 1173 and 184 doesn’t just cost you one batch. It costs you the rework, the customer complaint, and the credibility. I’ve spent seven years supplying photoinitiators to UV coating, ink, adhesive, and 3D printing resin manufacturers across Europe, India, and Southeast Asia. This guide gives you what the data sheets don’t: a real decision framework, grounded in production reality.
What Are Photoinitiator 1173 and 184?

Both belong to the Norrish Type I alpha-hydroxy ketone class — the most widely used free-radical photoinitiator category in UV curing. Under UV light, each undergoes unimolecular C–C bond cleavage adjacent to the carbonyl group, generating two reactive radical fragments that initiate polymerization of acrylate and methacrylate systems.
| Property | Photoinitiator 1173 | Photoinitiator 184 |
|---|---|---|
| Chemical name | 2-Hydroxy-2-methyl-1-phenyl-1-propanone (HMPP) | 1-Hydroxycyclohexyl phenyl ketone (HCPK) |
| CAS Number | 7473-98-5 | 947-19-3 |
| Trade names | Darocur 1173, Omnirad 1173, Speedcure 73 | Irgacure 184, Omnirad 184 |
| Physical form | Colorless to pale yellow liquid | White crystalline solid / powder |
| Melting point | ~4°C (liquid at room temperature) | 47–49°C |
| Boiling point | ~250°C at 760 mmHg | Decomposes above 150°C |
| Density | 1.074 g/cm³ at 20°C | ~1.19 g/cm³ |
| Refractive index | 1.502–1.504 at 20°C | N/A (solid) |
| Purity (typical) | ≥98.5% by GC | ≥99% by GC |
| Water solubility | ~13.3 g/L at 20°C | Practically insoluble |
| Flash point | ~122°C | >150°C |
| Shelf life | 12 months (sealed, dark, ≤25°C) | 24 months (sealed, dark, dry) |
| Standard packaging | 25 kg or 200 kg plastic drum | 25 kg cardboard drum |
| Shipping class | Combustible liquid (confirm with freight forwarder) | Standard chemical cargo |
The names “Darocur 1173” and “Irgacure 184” are legacy BASF/Ciba brand designations. Both molecules are now off-patent, produced by multiple manufacturers in China, and available under generic trade names with full CoA documentation.
The Core Technical Difference: Absorption, Form, and Radical Behavior
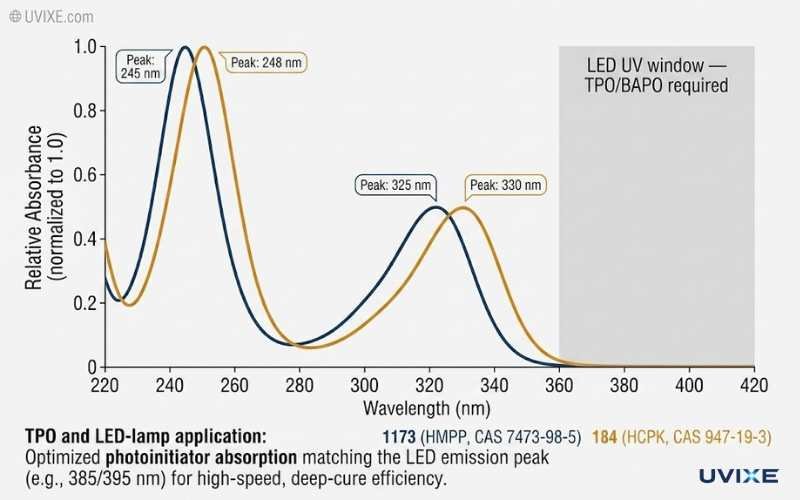
UV Absorption Spectra
Both photoinitiators absorb primarily in the UVB region (280–340 nm), which aligns well with the output of conventional medium-pressure mercury arc lamps.
- 1173 (HMPP): Absorption peaks at approximately 244 nm, 278 nm, and 330 nm. Primary photocleavage at 320–335 nm.
- 184 (HCPK): Strong absorption at 246–250 nm and a secondary peak at 320–335 nm. Slightly stronger short-wave absorption than 1173.
In practice, their cure performance under a standard mercury lamp is nearly identical within the same formulation. The performance divergence comes from physical form, production process, and downstream application — not spectroscopy.
According to a RadTech study on multiple photoinitiator systems, both HMPP (1173) and HCPK (184) belong to the alpha-hydroxy ketone class and respond similarly under mercury lamp output. The meaningful spectral gap opens only when you add phosphine oxides (TPO, BAPO/819) that cover 360–410 nm — a range neither 1173 nor 184 addresses.
Oxygen Inhibition: The Surface Cure Problem Both Share
Atmospheric oxygen scavenges free radicals before they can initiate polymerization, leaving a tacky, under-cured surface. This is a category-level constraint, not a 1173-vs-184 difference. Both are equally susceptible.
Standard mitigation strategies:
- Add an amine synergist (e.g., MDEA or EDAB) at 1–2% w/w
- Blend with a Type II initiator (ITX or benzophenone) that works via hydrogen abstraction
- Use nitrogen inerting over the cure zone
- Increase UV dose — higher lamp intensity or slower line speed
Running either photoinitiator without one of these strategies is a formulation risk, not a cost-saving measure.
Physical Form: The Real Production Differentiator
This is the decision point that matters most at the plant floor — and it’s rarely covered in competitor articles.
1173 (liquid):
- Disperses immediately at room temperature into acrylate systems
- No pre-heating, no dissolution step, no lag time
- Easier to dose by weight or volume pump in continuous mixing
- Functions as a liquid carrier for solid co-initiators (819, TPO) — critical for formulation stability
- Higher volatility: benzaldehyde photolysis byproduct is more pronounced — ensure adequate ventilation
184 (solid):
- Requires pre-dissolution in a reactive monomer (e.g., HDDA, TPGDA) or gentle warming before incorporation
- Lower volatility — production odor is less of an issue
- Better thermal stability in high-temperature compounding processes
- Longer shelf life under standard storage — more suitable for slow-moving inventory
- Recommended for UV coatings requiring prolonged sunlight exposure — slightly better weathering profile
Application Decision Matrix: When to Use 1173 vs 184
| Application | Substrate | Recommended | Reason |
|---|---|---|---|
| Clear UV wood coating | Parquet, furniture | 1173 or blend | Liquid integration; standard clear system |
| UV plastic coating | ABS, PC, PET | 1173 | Fast dispersion; low loading needed |
| UV paper overprint varnish | Paper, cardboard | 184 or blend | Solid form suits high-speed OPV lines; better long-term stability |
| Lithographic offset ink | Paper, board | 184 + ITX | Standard in offset systems; ITX addresses surface cure |
| Flexographic ink | Film, paper | 1173 | Liquid preferred in flexo formulations |
| UV screen printing ink | Plastics, textiles | 1173 + 184 blend | Blend maximizes solubility of co-initiators |
| UV structural adhesive | Metal, glass | 1173 | Liquid integration; no dissolution step in two-part systems |
| UV label / PSA adhesive | Film | 184 | Solid stability in pressure-sensitive systems |
| SLA/DLP 3D printing resin | — | 1173 + 819 | Liquid critical for resin homogeneity; 819 handles through-cure at LED wavelengths |
| UV LED curing (365/395 nm) | Any | Add TPO-L | Both under-absorb in LED range; phosphine oxide mandatory |
| White pigmented UV coating | Metal, plastic | 184 + TPO | 184 for surface; TPO for through-cure in TiO₂-loaded systems |
| Outdoor UV coating | Metal, wood | 184 | Better weathering resistance under prolonged UV/sunlight exposure |
My Field Observation on SLA/DLP Resins

In 3D printing resin formulations, 1173 is almost universally preferred over 184 — and not just because it dissolves instantly. DLP printers running at 405 nm often combine 1173 with 819 (BAPO) or TPO-L to extend absorption into the visible/near-UV range. Solid 184 creates dispersion challenges in the low-viscosity resin bases used for SLA. I’ve seen batches where undissolved 184 particles settled between print runs and caused curing inconsistency layer-to-layer — a defect that’s nearly impossible to diagnose without knowing where to look. With 1173, that failure mode doesn’t exist.
Dosage, Concentration, and Film Thickness Parameters
The most common mistake I see from new procurement buyers: assuming higher PI loading equals better cure. Above a critical threshold, excess photoinitiator actually blocks UV light from penetrating the film, resulting in poor through-cure even when the surface appears dry. This is called the “inner filter effect.”
The data below is based on our standard formulation testing protocol: 50 µm draw-down films on glass, cured under a 120 W/cm medium-pressure mercury lamp, conversion rate measured by FTIR at 810 cm⁻¹ (acrylate double bond consumption). These ranges align with published guidelines from Bomar Specialties and Longchang Chemical.
| Film Thickness | Recommended Dosage (1173 or 184) | Key Consideration |
|---|---|---|
| 2–5 µm (ultra-thin) | 3–5% w/w | Higher loading needed; very short optical path |
| 5–20 µm (thin) | 2–4% w/w | Standard clear coating; most OPV and plastic coating work |
| 20–50 µm (medium) | 1–3% w/w | Consider adding TPO/819 for reliable through-cure |
| 50–200 µm (thick) | 0.5–2% w/w | Long-wave PI mandatory; 1173/184 handles surface only |
| >200 µm (deep cure) | <1% 1173/184 + 1–2% TPO or 819 | 1173/184 contribution is surface only; phosphine oxides carry the depth |
Always run a ladder study before locking a formula — vary PI content from 0.5% to 5% in 0.5% increments and measure surface tack, pendulum hardness, and yellowing index (ΔYI) at each point. The optimal loading sits at the inflection point before through-cure begins to drop.
1173 + 184 Blend: The Synergy Most Suppliers Don’t Explain
One of the most actionable insights from real formulation work: blending 1173 and 184 is not just acceptable — it unlocks properties neither achieves alone.
A patent-documented finding (WO2002088192A1) shows that liquid 1173 alone can solubilize approximately 20% by weight of solid BAPO (819). When you add roughly equal weight of solid 184 to the liquid 1173, the mixture solubilizes 25% or more of BAPO — a property neither component achieves independently.
Why does this happen? The liquid 1173 acts as the primary solvent, while 184 alters the polarity and solvation shell of the mixture — creating a microenvironment that accommodates more of the bulky phosphine oxide structure of 819. This is not just a physical blending trick. It’s a formulation design principle: the 1173/184 pair becomes a higher-capacity carrier system for your co-initiators.
The same patent demonstrates that formulation stability also improves — blends of 1173 + 184 resist crystallization at lower temperatures compared to either component alone.
A common commercial product — the “PowerCure 1000” blend from Tintoll — is 80% 1173 + 20% 184 by weight, specifically designed for clear UV coating and ink systems where both surface and through-cure matter.
Recommended blend ratios:
| Blend Goal | 1173:184 Ratio | Application |
|---|---|---|
| General clear coating (easy handling) | 80:20 | Paper, plastic, wood overprint |
| Maximize 819/BAPO solubilization | 50:50 | High-performance clear coatings, 3D resins |
| Surface-cure emphasis | 70:30 | Thin-film OPV |
| Through-cure emphasis | Add TPO/TPO-L to either | Thick coatings, pigmented systems |

LED vs Mercury Lamp: The Purchasing Decision Most Teams Get Wrong
If your production line is converting to UV LED — or your customers are asking about LED compatibility — here is the one fact that changes your entire PI selection:
Neither 1173 nor 184 cures efficiently under a 365 nm or 395 nm LED source alone.
Both absorb primarily below 340 nm. LED sources at 365 nm and 395 nm output virtually no energy in that range. According to application data from Gansu Jindun Chemical, the spectral overlap between these photoinitiators and common LED emission wavelengths is minimal — meaning most lamp energy is wasted and radical generation is insufficient for reliable cure.
What to do by LED wavelength:
| LED Wavelength | Primary PI | Role of 1173/184 |
|---|---|---|
| 365 nm | TPO (absorbs 360–400 nm) | Keep at 0.5–1% for residual short-wave assist |
| 385 nm | TPO or TPO-L | 1173/184 at 0.5–1% max |
| 395/405 nm | TPO-L or 819 (BAPO) | 1173/184 contribution is marginal — reduce or eliminate |
This is not a failure of 1173 or 184. They were engineered for mercury lamp systems. Procurement teams ordering them as the primary initiator for LED lines are not getting the cure performance they expect — and the line speed penalties are real.
If your customers are asking about LED UV compatibility, our TPO-L product page at UVIXE includes absorption spectra and formulation starting points for 365 nm and 395 nm systems. Our photoinitiator 819 (BAPO) is the deepest-absorbing initiator in our portfolio and the standard recommendation for 405 nm LED cure.
Cost, MOQ, Sourcing, and Shipping Reality
Current Market Pricing (China Export, 2025 Reference)
| Product | Price Range (USD/kg) | Typical MOQ | Standard Packaging |
|---|---|---|---|
| Photoinitiator 1173 | $2–7/kg | 25 kg (1 drum) | 25 kg or 200 kg plastic drum |
| Photoinitiator 184 | $2–5/kg | 25 kg (1 drum) | 25 kg cardboard drum |
Volume discounts are significant: at 1,000 kg+, expect 30–40% reduction from spot price. Specialty or high-purity grades (low-migration, GC ≥99.5%) carry 20–50% premiums and are worth the cost for food-adjacent or cosmetic applications.
CoA Verification Checklist (What to Demand From Every Supplier)
Before accepting any shipment, your Certificate of Analysis must confirm:
- Assay (purity): ≥98.5% for 1173, ≥99% for 184 — by GC or HPLC
- Appearance: Clear pale yellow liquid (1173) / white powder or flakes (184) — no discoloration or haze
- Water content: <0.1% w/w — moisture causes clumping in 184 and hydrolysis risk in 1173
- Refractive index (1173 only): 1.502–1.504 at 20°C — a fast proxy purity check
- Melting point (184 only): 47–49°C — deviation indicates contamination or degradation
- Heavy metals: Below regulatory threshold for your target market (REACH, RoHS, FDA 21 CFR where applicable)

Shipping and Lead Time Notes
- 1173: Ships as combustible liquid; flash point ~122°C. Confirm UN classification with your freight forwarder by destination country — EU and India treat this differently.
- 184: Ships as standard chemical cargo; no UN hazmat classification in most jurisdictions.
- Lead time from China: 7–15 days ex-works; 25–35 days CIF Europe depending on port and routing.
- EU import: Confirm REACH pre-registration or registration status with supplier before order — both 1173 and 184 are registered under REACH.
REACH, Migration Limits, and Regulatory Compliance
Both photoinitiators are registered under EU REACH and widely used in regulated industries. But there are application-specific limits that matter — especially if your customers are in food packaging, toys, or cosmetics.
EU food-contact compliance (Regulation 10/2011):
Photoinitiators in food-contact plastics are subject to specific migration limits (SML). The European Food Safety Authority (EFSA) evaluates these. Photoinitiator 1173 has attracted particular scrutiny — the Swiss Federal Office of Public Health flagged 1173 and related alpha-hydroxy ketones for migration into food products through packaging as far back as 2012. Migration values for 1173 in LDPE films at 40°C/10 days have been measured at levels exceeding 0.01 mg/kg food simulant thresholds.
Practical guidance by application:
| Application | 1173 | 184 | Recommendation |
|---|---|---|---|
| Non-food industrial coating | ✅ Acceptable | ✅ Acceptable | Standard use |
| Outdoor UV coating | ✅ Acceptable | ✅ Preferred | 184 for better weathering |
| Food packaging (primary) | ⚠️ High migration risk | ⚠️ Lower but still evaluate | Use polymeric PI or verify SML compliance |
| Toy coatings (EN 71) | ⚠️ Verify | ⚠️ Verify | Test for extractables |
| Cosmetic UV nail gel | ✅ Common use | ✅ Common use | Confirm EU Cosmetics Regulation compliance |
SVHC status: Neither 1173 nor 184 is currently on the REACH SVHC candidate list as of 2025. Verify with your supplier for updates — the list is updated twice yearly.
Worker exposure: Both generate odorous photolysis byproducts — benzaldehyde from 1173, cyclohexanone and benzaldehyde from 184. Ensure adequate exhaust ventilation in production. Full SDS in GHS format (English + local language) is required for EU, Indian, and Southeast Asian customs declarations.
FAQ
Is photoinitiator 1173 the same as Darocur 1173?
Yes. “Darocur 1173” is the original BASF/Merck trade name. The active molecule is 2-hydroxy-2-methyl-1-phenyl-1-propanone, CAS 7473-98-5. Today it is manufactured by multiple producers globally under names including Omnirad 1173, Speedcure 73, and generic PI-1173. The chemistry is identical across brands. What varies between suppliers is purity, batch-to-batch consistency, and packaging format — all verifiable by CoA. Always request GC assay data, not just a certificate of conformity.
Can I replace 184 with 1173 directly in my formula without retesting?
No — and this is a mistake I see regularly. Beyond the obvious physical form difference (184 requires pre-dissolution, 1173 does not), the two molecules have slightly different initiation kinetics in specific oligomer systems. In a urethane acrylate system at 50 µm film thickness under 120 W/cm mercury lamp, we’ve measured 3–8% variance in double bond conversion rate between identical loadings of 1173 and 184. That may be acceptable. It may not be. Always run a minimum FTIR conversion check and a pendulum hardness test before signing off on a formula change — even a “like-for-like” substitution.
Which one is better for non-yellowing UV coatings?
Both are among the lowest-yellowing Type I photoinitiators available. A yellowing index comparison study by Longchang Chemical using pentaerythritol triacrylate propoxide as the resin base places 1173 and 184 in the same low-ΔYI tier alongside 754 and MBF — well below photoinitiators containing amine-substituted aromatic structures. For outdoor UV coatings exposed to prolonged UV radiation, 184 shows marginally better weathering stability in our aging chamber tests (500h QUV-A), likely due to its higher molecular weight and lower volatility post-cure. For both applications, pair with a HALS stabilizer (e.g., TINUVIN 292 or equivalent) to extend color stability beyond 12 months.
What is the optimal blend ratio of 1173 and 184?

The 80:20 (1173:184) ratio by weight is the most widely used commercial starting point — it gives a liquid-dominant blend that disperses easily while retaining the solubility benefits of 184. For applications where you also need to dissolve significant amounts of 819 or TPO, a 50:50 ratio is more effective — the blended solvent system accommodates 25%+ of solid BAPO versus ~20% with 1173 alone. This is documented in patent WO2002088192A1. Adjust within this range based on your system viscosity and production mixing temperature.
Do I need a co-initiator with 1173 or 184?
Almost always, yes. Both suffer from oxygen inhibition in ambient-atmosphere curing — if your cure zone is not nitrogen-inerted, you will get a tacky surface layer regardless of PI loading. The fix: add an amine synergist (MDEA, EDAB, or EDB) at 1–2% w/w, or blend with a Type II initiator (ITX, benzophenone) that provides hydrogen abstraction. For through-cure in films above 50 µm, add TPO or BAPO (819) at 0.5–1.5%. In our testing, adding 1% MDEA to a 2% 1173 system improved surface cure conversion from 61% to 89% RAU under identical lamp conditions. That gap is not recovered by simply increasing PI loading — the oxygen problem requires a chemical solution, not a concentration solution.
Stop Guessing — Your Line Speed and Reject Rate Depend on This Call
1173 and 184 are not generic twins. The physical form difference alone changes your production workflow. The migration behavior changes your regulatory exposure. The LED compatibility gap changes your capex planning if you’re switching lamp systems.
Get those three variables right — form, migration risk, lamp type — and you’ve made 90% of the decision. The remaining 10% is in the dosage ladder study. There is no shortcut for that last step, and any supplier who tells you otherwise is selling you convenience over quality.
If you’re evaluating both photoinitiators for a new formulation, the fastest path to a correct answer is a small sample test at three loading levels (1%, 2%, 4%) under your actual production lamp. We can help you design that test.
Talk to a Supplier Who Can Read a CoA and a Formulation at the Same Time
At UVIXE, we supply photoinitiator 1173 and photoinitiator 184 with full batch CoA, GC assay data, and SDS documentation as standard. Our product line also covers TPO, TPO-L, 819 (BAPO), ITX, and DETX — so when your formulation needs a co-initiator or a LED-compatible phosphine oxide, you’re not sourcing from three different suppliers.
Three ways to engage:
- Request a sample — 100–500 g sample with CoA for lab evaluation
- Get a formal quote — specify volume, packaging, and delivery destination for a complete landed-cost quote with lead time
- Technical consultation — share your application and current formula; we’ll recommend grade, loading, and blending strategy before you commit to an order
MOQ: 25 kg per product. Bulk pricing at 500 kg+. Standard lead time: 7–15 days ex-works China.



